For decades, scientists have known our bodies have an “inner cannabis system” – the endocannabinoid system (ECS). It’s a master regulator involved in everything from memory and mood to pain and appetite. We knew the key players: receptors like CB1, and our body’s own cannabis-like molecules, 2-AG and anandamide (AEA).
But a major challenge has been this: endocannabinoids are like fleeting whispers in the brain, appearing and disappearing in seconds to minutes. How do they actually work when we’re awake, moving, learning, or even during a seizure? What’s really happening at specific synapses?
A groundbreaking review in the prestigious journal Neuron by Malhotra et al. (2025) highlights how new technologies are finally letting us see the ECS in action in behaving animals, with stunning precision. This isn’t just about cool science; it has profound implications for understanding health, disease, and how cannabis-based medicines might work.
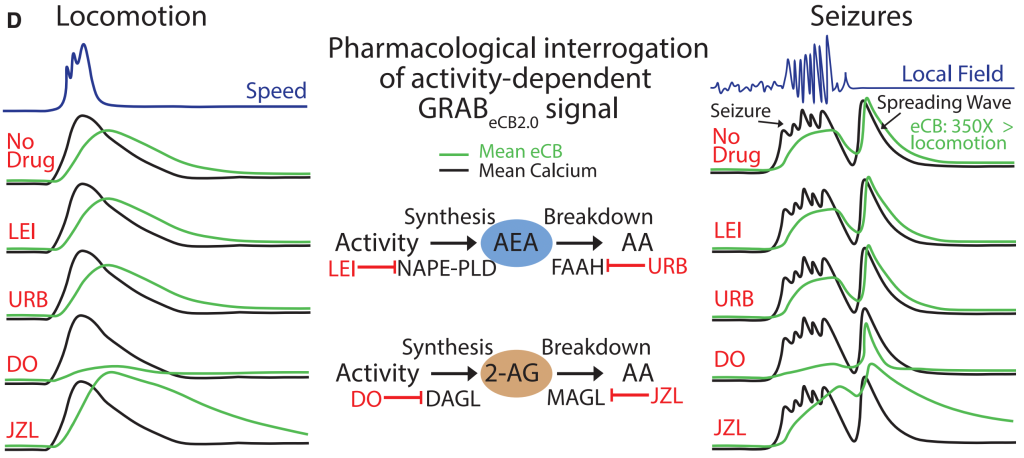
Novel sensor technology, as shown in this adapted figure from Malhotra et al. (2025), allows direct visualization of endocannabinoid release in behaving mice. During both normal locomotion and seizures, the rapid endocannabinoid signal (green) is almost entirely driven by 2-AG. Manipulating anandamide (AEA) pathways did not affect these activity-dependent signals, highlighting 2-AG’s primary role.
(Adapted from Malhotra et al.,, Integrating endocannabinoid signaling, CCK interneurons, and hippocampal circuit dynamics in behaving animals, Neuron (2025), https://doi.org/10.1016/j.neuron.2025.03.0.) Licensed under CC BY 4.0.
Key Breakthroughs Unveiled
2-AG Takes Center Stage (In Vivo)
For years, there’s been debate about whether 2-AG or AEA is the primary “on-demand” endocannabinoid. Using a novel fluorescent sensor (GRABeCB2.0), researchers can now directly visualize endocannabinoid release in real-time in the brains of active mice.
The verdict? During normal brain activity (like locomotion) and even during seizures, the rapidly changing signal is overwhelmingly 2-AG. Blocking 2-AG production stops the signal; blocking its breakdown prolongs it. Manipulating AEA, on the other hand, had little effect on these rapid, activity-driven signals.
This pinpoints 2-AG as a critical target. Therapies aiming to modulate acute, activity-dependent ECS responses (e.g., in epilepsy, or for certain aspects of learning and memory) might benefit from focusing on 2-AG pathways (synthesis or degradation enzymes like MAGL).
Surgical Precision of ECS Signaling
The 2-AG signals are incredibly precise. They are released locally by active neurons and act on nearby nerve terminals, typically within seconds and confined to tiny spaces.
This isn’t a broad, system-wide “wash” of endocannabinoids. It’s a highly specific, “only when and where needed” form of communication.
Why this matters: It reinforces that the ECS is about fine-tuning. When considering medical cannabis, this precision highlights why different cannabinoids with different actions (e.g., THC as a direct CB1 activator vs. CBD which modulates ECS tone indirectly) might have such diverse effects. It also points to the challenges and opportunities in developing targeted ECS therapies.
The ECS at Work During Normal Behavior – Shaping Memory & Navigation
A long-standing question was whether the ECS is only recruited by strong, artificial stimuli or if it’s active during everyday brain function.
The new research shows that normal activities, like an animal navigating its environment, generate enough neuronal activity to release 2-AG.
Focus on DSI In Vivo: A process called Depolarization-induced Suppression of Inhibition (DSI), where 2-AG release from an active neuron temporarily quiets down incoming inhibitory signals, was directly observed in behaving animals. This was shown to be crucial for the precise function of “place cells” in the hippocampus, which are essential for spatial memory. Mice lacking CB1 receptors on certain inhibitory neurons (CCK interneurons, the main targets for 2-AG in DSI) had fuzzier spatial maps.
Why this matters: This directly links ECS (2-AG/CB1) function to cognitive processes like learning and memory formation. It provides a cellular mechanism for why cannabinoids can impact these functions and why ECS dysregulation is implicated in cognitive disorders.
ECS Overdrive in Seizures – A Double-Edged Sword
During acute seizures, the brain is flooded with a massive (over 100-fold) surge of 2-AG. This signal is widespread and decays within a minute after the seizure stops.
This 2-AG surge can be neuroprotective (activating CB1 to dampen excitability). However, because 2-AG breakdown produces arachidonic acid (AA), the precursor for inflammatory prostaglandins, this massive release can also fuel post-seizure inflammation and even brain hypoxia (lack of oxygen). This is the “Janus-faced” nature of 2-AG in pathology.
Why this matters: This is critical for understanding epilepsy and the therapeutic potential of cannabinoids. Modulating MAGL (the enzyme that breaks down 2-AG) could both boost the protective CB1 signaling and reduce harmful prostaglandin production. This explains why MAGL inhibitors are a hot area of research.
What About Anandamide (AEA)?
The review notes that while the new sensors can detect AEA, the fast, activity-dependent signals observed in vivo so far are dominated by 2-AG.
This doesn’t mean AEA isn’t important. It might have different roles – perhaps slower, more “tonic” (background) signaling, or be more prominent in other brain regions or under different conditions. Its synthesis enzyme (NAPE-PLD) is found in different locations, suggesting distinct functions.
Why this matters: The ECS is not monolithic. Different endocannabinoids likely have specialized roles. This reinforces the need for nuanced approaches in ECS-targeted therapies.
The Bigger Picture for Healthcare & Cannabis Medicine
This new wave of in vivo research, beautifully summarized by Malhotra et al., is transforming our understanding of the ECS from a collection of molecular components into a dynamic, functioning system observable in real-time.
For HCPs: It underscores why the ECS is so fundamental to brain health and disease. Recognizing the precision and activity-dependence of 2-AG signaling can inform how we think about neurological and psychiatric conditions where the ECS is implicated (autism, anxiety, epilepsy, Alzheimer’s, etc.).
For Medical Cannabis Professionals: It provides deeper insight into how cannabinoids might be working. Understanding that phytocannabinoids like THC interact with a system that naturally uses 2-AG for highly precise, localized signaling helps explain the complexities of dose, effect, and individual variability. It also highlights why non-THC cannabinoids like CBD, which modulate ECS tone or target other receptors, can have distinct therapeutic profiles.
The ability to see the ECS at work in behaving animals opens up incredible avenues for developing more targeted and effective therapies for a vast range of conditions. The future of ECS science is not just about understanding molecules, but understanding their dynamic interplay in the living, working brain.
Reference:
Malhotra S, Donneger F, Farrell JS, Dudok B, Losonczy A, Soltesz I. Integrating endocannabinoid signaling, CCK interneurons, and hippocampal circuit dynamics in behaving animals. Neuron. Published online April 19, 2025. doi:10.1016/j.neuron.2025.03.016

